Szilvia Juhász, Benjamin Tamás Papp, Anna Tácia Fülöp, Zoltán Farkas, Dávid Kókai, Dóra Alexandra Gyémánt, Franciska Tóth, Zsófia Nacsa, Dóra Spekhardt, Balázs Koncz, Péter Burkovics, Csaba Pál & Máté Manczinger
Cancer cells carry thousands of mutations, but not all mutations are created equal. Some make tumors highly visible to the immune system, while others help cancers hide. In this study, we discovered that across thousands of human cancers, there are five dominant patterns of protein-altering mutations — called amino acid substitution signatures — and these patterns help determine how tumors interact with the immune system.
When DNA in a cell is damaged by environmental exposures (such as tobacco smoke or UV light) or by internal errors during replication and repair, the resulting mutations alter the building blocks of proteins — amino acids. By analyzing nearly 9,300 cancer genomes from diverse cancer types, we found that instead of a random jumble of changes, nearly all tumors are dominated by one of five characteristic substitution signatures.
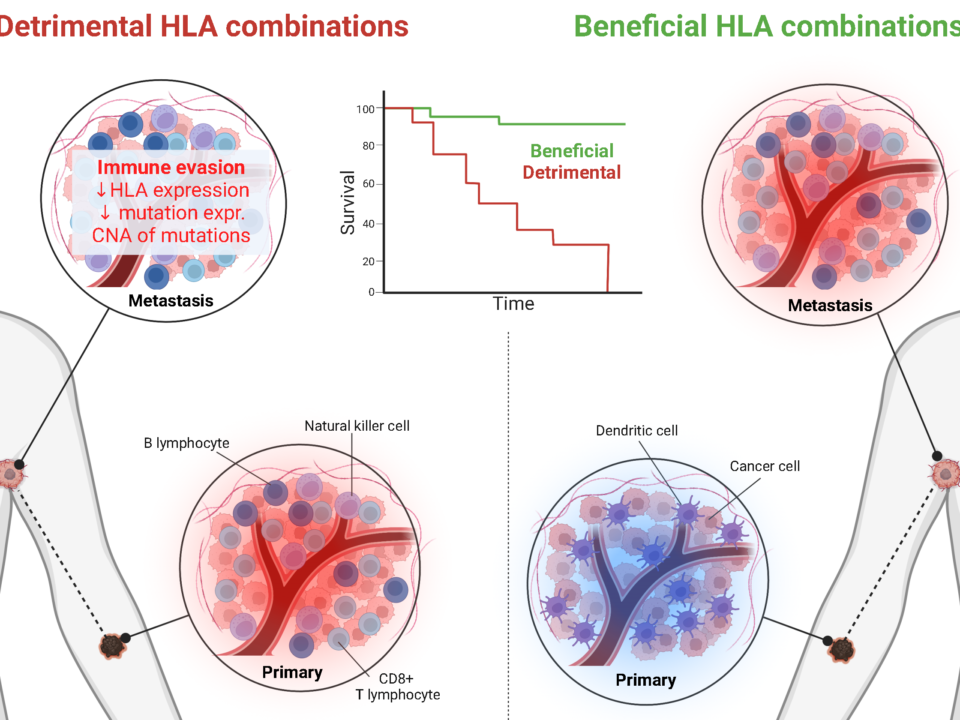
Crucially, these five signatures are not only molecular fingerprints of how the mutations arose — they also influence how well the immune system “sees” the tumor. Some mutation patterns tend to create highly immunogenic protein fragments (neoantigens) that alert immune cells, while others produce less recognizable neoantigens, leading to “cold” tumors that evade immune attack.
“Despite the diversity of mutational processes, their protein-level consequences converge into just five recurring fingerprints, which can strongly influence immune recognition,” we conclude. One of the most striking findings involves a mutational pattern linked to DNA repair defects and chemical exposures. Tumors dominated by this pattern often respond poorly to immune checkpoint inhibitor therapies, even when their overall mutational burden is high. In other words, a tumor can harbor many mutations and still generate too few effective immune targets.
“Mutational burden alone is insufficient. The qualitative, protein-level consequences of mutations are critical for understanding why immunotherapy fails in many patients,” we emphasize.
However, we also show that certain genetic variants in the human immune system — such as specific HLA class I types common in Europeans — can partially counteract this effect by more effectively presenting some of these mutated peptides to T cells. This suggests that the same tumor may be more immunologically visible in one patient than in another.
Taken together, our findings point toward a more refined framework for predicting immunotherapy response. Tumor visibility to the immune system is not determined by mutation numbers alone, but also by the protein-level patterns those mutations create. Our results support a new framework for truly personalized immunotherapy, integrating tumor genomics with the patient’s immunogenetic background.
Beyond scientific insight, our work also carries broader societal relevance. More accurate prediction of therapy response could help reduce unnecessary treatments, limit avoidable side effects, and shorten the time needed to identify effective therapies for individual patients.
The study was carried out through close collaboration between multiple research groups.
The program was funded by international and national grants.